
Malignant
Disease:
Special Procedures
Staging
of Gynecologic
Oncology Patients With
Exploratory Laparotomy
Subclavian Port-A-Cath
Peritoneal Port-A-Cath
Application
of Vaginal
Cylinders for Intracavitary
Radiation Therapy
Application
of Uterine Afterloading Applicators
for Intracavitary Radiation Therapy
Pelvic High-Dose
Afterloader
Abdominal
Injection of Chromic Phosphate
( ) )
Supracolic
Total Omentectomy
Omental Pedicle "J"
Flap
Tube Gastrostomy
Total Vaginectomy
Radical
Vulvectomy
With Bilateral Inguinal
Lymph Node Dissection
Reconstruction
of the
Vulva With Gracilis Myocutaneous Flaps
Transverse
Rectus
Abdominis Myocutaneous
Flap and Vertical Rectus
Abdominis Myocutaneous
Flap
Radical
Wertheim
Hysterectomy With
Bilateral Pelvic Lymph
Node Dissection and With Extension of the Vagina
Anterior Exenteration
Posterior Exenteration
Total Pelvic
Exenteration
Colonic
"J" Pouch Rectal
Reservoir
Kock Pouch
Continent Urostomy
Omental "J" Flap
Neovagina
Ileocolic
Continent Urostomy (Miami Pouch)
Construction
of Neoanus
Gracilis Dynamic Anal
Myoplasty
Skin-Stretching
System Versus Skin Grafting
Gastric
Pelvic Flap for
Augmentation of Continent Urostomy or Neovagina
Control
of Hemorrhage in Gynecologic Surgery
Repair
of the Punctured
Vena Cava
Ligation
of a Lacerated
Internal Iliac Vein and
Suturing of a Lacerated Common Iliac Artery
Hemorrhage
Control in
Sacrospinous Ligament
Suspension of the Vagina
Presacral
Space
Hemorrhage Control
What
Not to Do in Case of Pelvic Hemorrhage
Packing
for Hemorrhage
Control
Control
of Hemorrhage
Associated With Abdominal Pregnancy |
Kock Pouch Continent Urostomy
Patients who have lost the use of the bladder as a result of irradiation
or surgical excision may be candidates for a procedure involving the
construction of a continent ileal reservoir for cutaneous urinary diversion.
The Kock pouch, designed by Nils Kock in 1982, was devised as a continent
urostomy. Modification by Donald Skinner provided a urinary diversion
as a continent nonrefluxing urostomy. This alternative to urinary diversion
deserves the consideration of gynecologic oncologist.
Physiologic Changes. Skinner et al. have shown that
construction of an internal reservoir suitable for urinary bladder
placement must provide for (1) retention of 500-1000 mL of fluid, (2)
maintenance of low pressure after filling, (3) elimination of intermittent
pressure spikes, (4) true continence, (5) ease of catheterization and
emptying, and (6) prevention of reflux.
The ileal mucosa of the pouch
appears to adapt well to urine; villus height decreases, and in time,
the mucosa becomes nearly flat, thereby reducing the absorption of
electrolytes from the urine.
Points of Caution. Prerequisites to construction
of the Kock pouch include reasonable renal function (creatinine less
than 3.0 mg/dL); adequate length of small bowel, so that utilization
of 80 cm of ileum will not result in a significant short bowel syndrome;
and a patient who is motivated for the procedure and who understands
the inherent risk (a 10-15% incidence of malfunction of the continent
valve mechanism, requiring additional surgery).
The low pressure within
the pouch and the high pressure within the nipples prevent reflux and
allow the patient to remain continent. In accordance with Laplace's
law, the low pressure within the pouch is maintained at high fluid
volumes.
A midline incision is preferred for construction of a continent
urostomy. The site of the stoma can be determined preoperatively. For
young, slim women we prefer to place the opening below the underwear
line immediately above the pubic hair. For older or obese patients,
the stoma is often placed higher to facilitate catheterization. The
surgeon should not feel bound by the preoperative stomal site marking,
however, if the mesentery does not allow the pouch to reach that location.
Since no appliance is worn for the collection of urine, the surgeon
need not worry about skin folds. When the pouch procedure is done in
conjunction with total pelvic exenteration or cystectomy, pelvic resection
is performed first. For conversion of an existing ileal conduit, all
the intra-abdominal wall adhesions must be taken down.
In summary, our
motivation for using the Skinner modifications of the Kock pouch continent
urostomy in gynecologic oncology has been 85% for medical reasons (i.e.,
to prevent contaminated reflux and thus deterioration of upper renal
units commonly associated with ileal or colonic loops) and 15% for
improvement in the quality of life. The elimination of the urinary
bag with its attendant problems of awkwardness and odor has a positive
effect on the quality of the patient's self-image and sexuality.
Technique
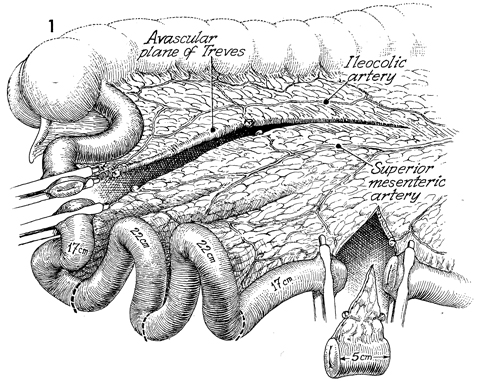
In this view the descending colon, cecum,
and terminal ileum, note that the avascular plane of Treves has
been entered after the terminal ileum was divided. The incision
in the avascular plane of Treves was carried lateral to the ileocolic
artery and medial to the superior mesenteric artery. The blood
supply of the entire Kock pouch depends on the superior mesenteric
artery and its branches. A 5-cm plug of ileum with its mesentery
is removed at the upper limit of the Kock pouch to allow placement
of the efferent limb of bowel at the preferred stoma site. The
first 17-cm segment of the pouch is marked off and labeled for
the efferent limb of bowel and the efferent nipple. The next
22-cm segment comprises one loop of the U-shaped pouch, and the
last 22-cm segment forms the other limb of the U-shaped loop
of the pouch. The last 17-cm segment is available for the afferent
nipple and bowel limb of the pouch. This segment is not necessary
if the surgeon is converting an ileal loop to the pouch. |
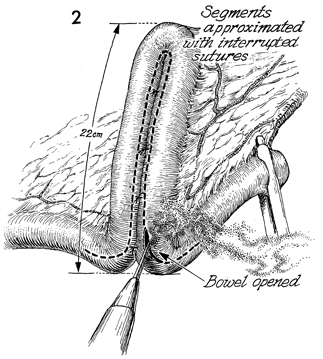
The two 22-cm U-shaped limbs are placed adjacent
to each other, and interrupted 3-0 polyglycolic acid (PGA) sutures
are placed in the bowel 1 cm above the junction of the two segments.
A cautery is used to open the intestine approximately 2 cm from
the junction of the mesentery, as indicated by the dotted
line. This opening is extended on the efferent and afferent
limbs for a distance of approximately 5 cm; the cautery electrocoagulates
the small blood vessels on the edge of the bowel. |
A 3-0 PGA suture on a straight fine intestinal
needle runs through the back wall of the pouch. A second layer
of 3-0 PGA sutures is placed between these sutures. |
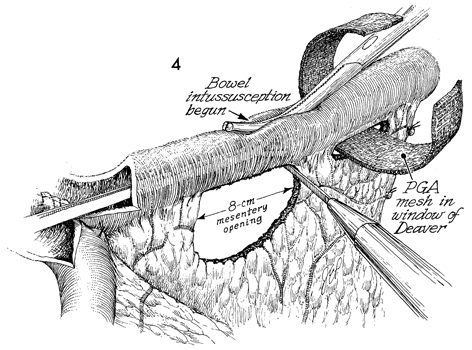
Construction of the nipples
in the afferent and efferent limbs has begun. An 8-cm opening
is created in the mesentery by opening the windows of Deaver,
applying Hendren's rule that 4 cm of mesentery adjacent to the
small bowel can be undercut because there is enough lateral vasculature
in the small bowel wall to prevent necrosis. An opening of 8
cm is essential for nontraumatic intussusception and to prevent
"extussusception," i.e., undoing of the intussusception. A Babcock
clamp is inserted into the lumen of the bowel, and a small Kelly
clamp is used to indent the bowel wall into the Babcock clamp.
Papaverine, 300 mg in 500 mL
of normal saline, must be administered intravenously 10-15 minutes
before intussusception for smooth muscle relaxation to allow
intussusception without trauma to the small bowel. A slight drop
in the patient's blood pressure should be expected; this rarely
exceeds 20 mm Hg in systolic pressure and 10 mm Hg in diastolic
pressure.
A 2-cm strip of PGA mesh is
passed through the window of Deaver. This will be sutured in
place after the nipple has been created. |
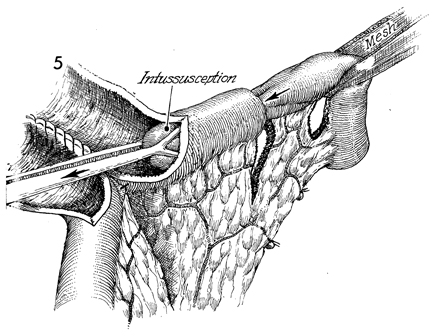
The intussusception is being performed under
the influence of a smooth muscle relaxant. A segment of bowel
is pulled out for a distance of 6-7 cm. |
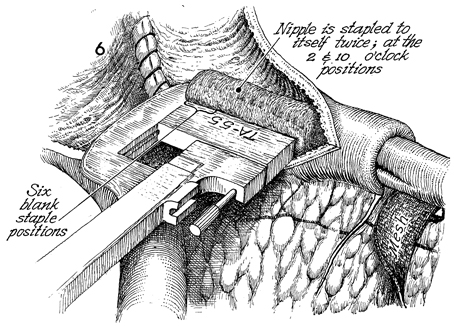
The TA-55 4.8-mm stapler
with 5 staples missing from the heel of the stapler is inserted
on the nipple and stapled at the 2 and 10 o'clock positions. |
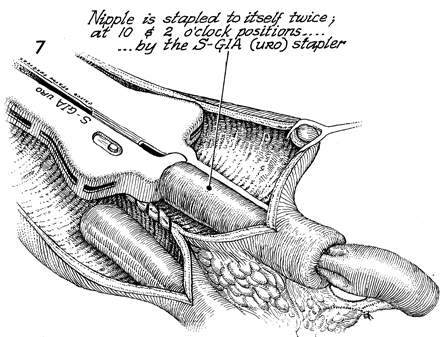
The nipple is stapled to itself
twice, at the 10 and 2 o'clock positions, with the S-GIA URO
stapler (United States Surgical Corp.). The stapler contains
no blade, and the inner rows of staples have been removed. It
has the advantage of having no pinhole. |
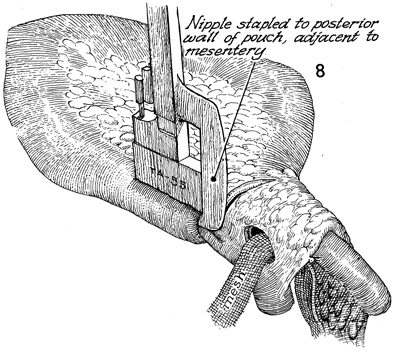
An alternative method would be to perform
a small enterotomy in the posterior wall of the pouch; place
the stapler through the enterotomy and then through the nipple,
and staple it. All pinholes from the TA-55 stapler must be sutured
with interrupted 3-0 PGA sutures before proceeding. Note that
the PGA mesh in the window of Deaver is still not sutured in
place. |
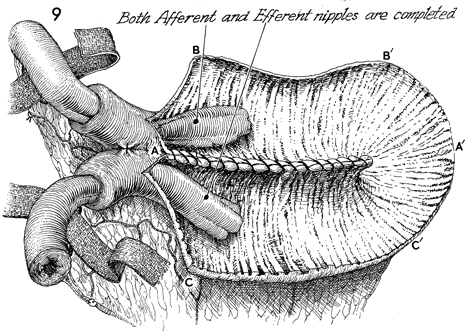
Both afferent and efferent nipples
have been completed. Note that the two strips of PGA mesh pass
through the windows of Deaver in the mesentery of both the efferent
and afferent bowel limbs adjacent to the nipples. The letters A to A', B to B',
and C to C' delineate the order of suture placement
that will produce a spherical rather than a tubular pouch. |
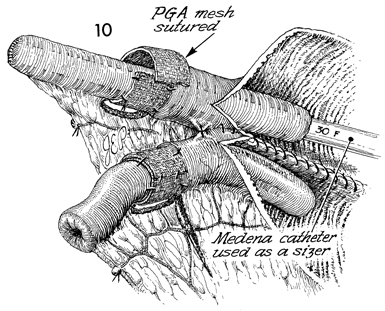
With the pouch still open, the No. 30 French
Medena catheter is inserted up the nipple in a retrograde fashion
to allow accurate sizing for the PGA mesh. The mesh is sutured
with interrupted 3-0 PGA sutures in a fashion that securely locks
it at the junction of the intussusception to prevent extussusception. |
|
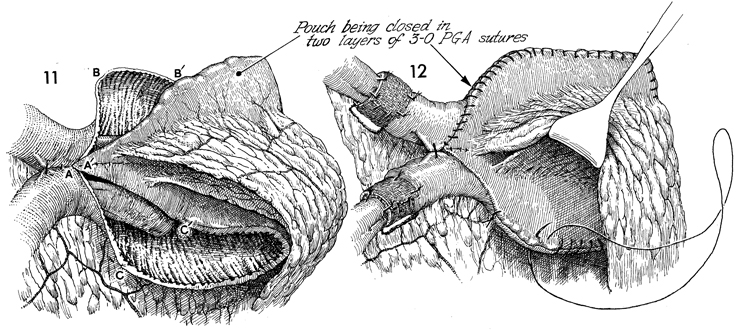
The
pouch is closed with 3-0 PGA sutures by approximating points A to A', B to B',
and C to C'. |
The remaining walls of the pouch are sutured
with a running 3-0 PGA suture, and a second layer of running
3-0 PGA sutures is added. |
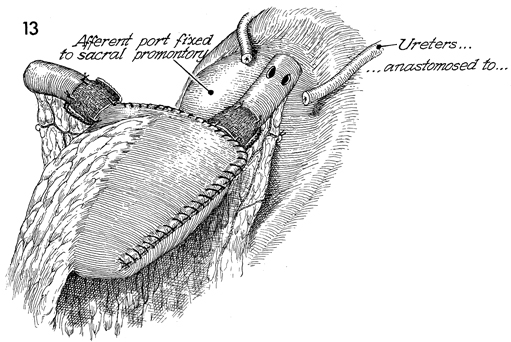
In the completed pouch, the PGA mesh is in
place adjacent to the intussusception. Two enterotomies have
been made in the afferent limb of the bowel. Two No. 8 French
Finney J Silastic catheters are threaded through the enterotomies,
down the afferent limb, through the afferent nipple, and into
the pouch. The ureters previously mobilized have been spliced
by incising the ureteral wall for a distance of approximately
3 cm. |
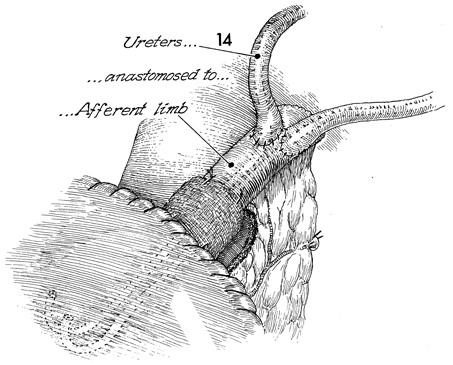
The Finney J Silastic catheter has been threaded
up the ureters and into the renal pelvis. The ureter has been
sutured to the enterotomy in the bowel with interrupted 4-0 PGA
sutures in a mucosa-to-mucosa technique. Additional sutures are
placed between the serosa of the bowel and the ureter. Indigo
carmine dye, 3 mL, is administered intravenously. The suture
line is thoroughly inspected to ensure that there is no leakage
of blue dye-stained urine coming from the kidney and down the
ureter. Note that the Finney J Silastic catheters are threaded
into a loop. The J curled ends of the catheters indicate they
are within the pouch and not in the nipple. |
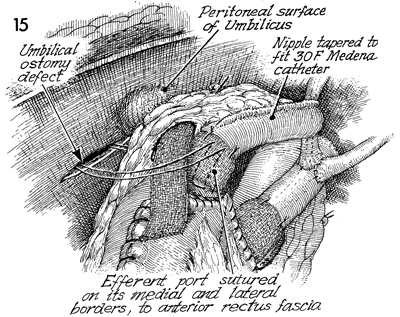
The efferent bowel limb has been exteriorized
through an umbilical ostomy defect. In addition, the efferent
bowel limb has been tapered to fit a No. 20 French Medena catheter
with the GIA instrument. Both of these procedures, exteriorization
through the umbilicus and the tapering of the efferent bowel
limb, are designed to reduce the diameter of the efferent bowel
limb and therefore raise the pressure of the overall efferent
system to greatly exceed that in the pouch.
The efferent port is sutured
on its medial and lateral borders to the anterior rectus fascia. |
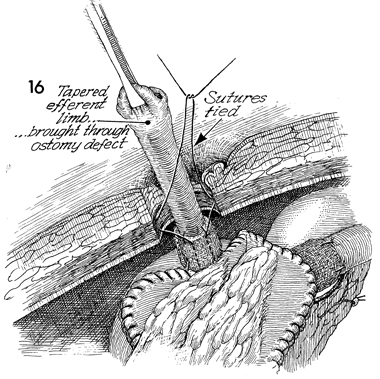
The tapered efferent bowel limb is brought
through the ostomy defect in the umbilicus. The sutures are tied
to the umbilical ostomy defect. |
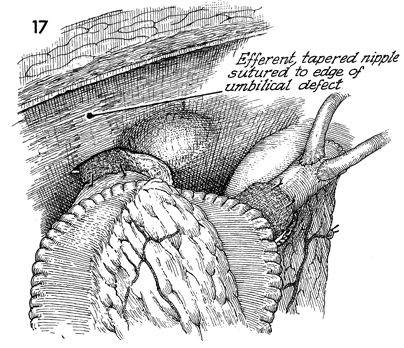
The efferent tapered nipple bowel limb is
sutured to the edge of the umbilical defect. |
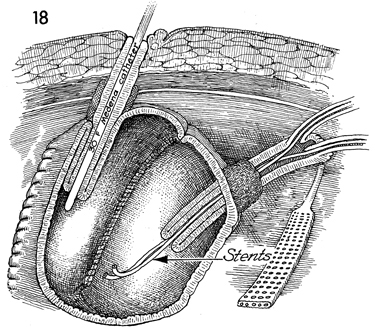
A No. 20 French Medena catheter is inserted
through the efferent bowel limb through the efferent nipple into
the pouch. The stoma has been matured in the umbilicus. The afferent
bowel limb containing the ureters and the afferent nipple are
shown on the right. Note the J Silastic stents coming from the
renal pelvis down the ureters through the afferent bowel limb
through the afferent nipple into the pouch. These are removed
with a cystoscope 3 weeks following surgery.
The very important Jackson-Pratt
suction drainage shown on the right remains in place until the
pouch has completely healed and there is no leakage from the
pouch or any of the anastomoses. This Jackson-Pratt drain is
usually removed 3 weeks postoperatively. |
|

















